When Breaking Builds: The Hidden Physics of organ Development
We grow up learning that breaking things is bad. A cracked vase is ruined. A fractured bone means weeks in a cast. But what if the very act of breaking were one of nature’s most elegant construction tools? A fascinating line of research now shows that mechanical forces in organ development — including controlled fracturing and microscopic cracks in tissue — help shape the complex organs that form in our bodies during embryonic growth. In other words, sometimes you have to break it to make it. What once seemed like destruction may actually be a powerful building strategy in biology.
The Builder’s Paradox: How Nature Uses Cracks to Craft Our Bodies?
Think about how you might assume a body is built. Cells divide, they stick together, they grow into shapes — a smooth, additive process, like sculpting clay by adding more and more material. And indeed, much of development works this way. But there is a problem: some of the structures in our bodies are far too intricate, too folded, too branched, to be explained purely by cells gently cooperating.
The elaborate folds of the brain, the delicate branching of the lungs, and the tiny finger-like projections lining the gut are astonishingly complex. These structures arise through processes studied in fields such as Morphogenesis, where scientists investigate how biological forms emerge during development.
Researchers are now discovering that part of the answer lies in something that sounds destructive: fracture. Not catastrophic breakage, but tiny, controlled structural failures guided by tissue mechanics and the physical properties of living matter… in just the right places, at just the right times. These fractures are not accidents. They are features — programmed into the physics of the tissue itself.
This idea reveals the surprising role of fractures in biology. Instead of representing damage, these microscopic breaks can help organize tissues and guide cells into forming functional structures.
Mechanobiology : How Living Tissue “Cracks” on Purpose
To understand this, imagine stretching a thin sheet of rubber from multiple directions. Eventually the stress becomes too great, and the sheet tears. The location and pattern of the tear depend on the sheet’s geometry, thickness, and the forces acting upon it. Physics determines where the crack will appear.
Now replace that rubber sheet with a layer of living cells — an epithelium — growing across a surface or being compressed by surrounding tissues. The same mechanical principles apply. Stresses build up, and when they exceed what the cellular sheet can withstand, tiny fractures or separations may occur.
The key difference is that living tissues can adapt. Cells can migrate, repair, and reorganize themselves around these disruptions. This is part of how cells respond to mechanical stress. Cells sense forces in their environment and alter their behavior accordingly….
A microscopic crack can guide cellular movement, influence growth patterns, and ultimately shape the architecture of an organ. A small separation in tissue can evolve into a fold, a branch, or a structural boundary that becomes essential for function.
Biomechanics and Mechanobiology: A New Lens on Old Questions
For decades, scientists believed that organ shapes were controlled almost entirely by ‘chemical signals’ called morphogens. These molecules diffuse through tissues and instruct cells about their identity and behavior.
But the emerging field of Mechanobiology shows that chemical signals alone cannot explain the complexity of biological structures. Instead, physical forces and biochemical signals work together… What the fracture research reveals is that *physics and chemistry work as partners*. Chemical signals may set the stage, but mechanical forces — including the forces that cause tissue to fracture — execute the performance.
This means that mechanical forces in organ development act alongside genetic programs to shape the body.
Consider the developing gut. As it grows, its inner lining becomes compressed and begins to buckle, much like a tablecloth that wrinkles when pushed from both ends. These ridges eventually become villi — the small finger-like projections that dramatically increase the intestine’s surface area for nutrient absorption.
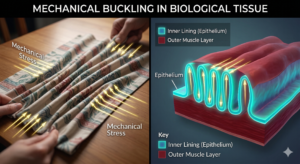
Researchers have shown that these structures arise partly from mechanical stresses within the tissue. Without these physical forces, the villi fail to form correctly, even when all the necessary molecular signals are present.
Forces Behind Branches and Folds
Similar principles appear in lung development. The lungs form an intricate branching network of airways that divide repeatedly, creating a tree-like structure that allows efficient gas exchange.
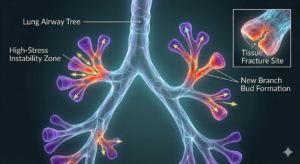
Mechanical instabilities and local separations within the tissue can help determine where new branches emerge. Over time, these small physical events contribute to a complex system of tubes that ultimately enables breathing.
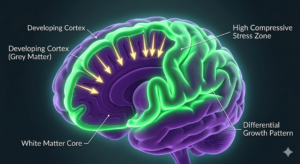
Brain development also reflects the influence of tissue mechanics. The brain’s iconic wrinkles, too, arise in part because the rapidly growing outer layer (the cortex) is mechanically constrained by the slower-growing tissue beneath it. The mismatch in growth rates creates compressive stress, and the cortex buckles — fractures — into its characteristic folds. Smooth-brained animals simply have less mismatch .
These folds increase surface area and enable greater neural complexity. In species with less growth mismatch between brain layers, the brain remains relatively smooth.
Why This Matters Beyond the Lab
Understanding how fractures and mechanical forces shape tissues has important implications for medicine and biotechnology.
-
Birth Defects and Disease
If mechanical forces in organ development are essential for shaping organs, then disruptions to tissue mechanics could lead to developmental abnormalities. Genetic mutations that alter cell stiffness, environmental toxins that affect tissue elasticity, or abnormal growth patterns might all interfere with the physical processes that guide organ formation.
This perspective suggests that some congenital disorders may arise not only from genetic mistakes but also from problems in the mechanical environment of developing tissues.
-
Tissue Engineering and Regenerative Medicine
Scientists working in regenerative medicine aim to grow replacement organs in the laboratory. While stem cells can be guided to become many different cell types, recreating the complex shapes of real organs remains extremely challenging.
Insights from tissue mechanics may offer a solution. Engineers might design scaffolds that intentionally introduce mechanical stresses or controlled instabilities, allowing tissues to fold and branch naturally.
In other words, future bioengineers may need to design structures that are meant to crack in specific ways — allowing controlled failures to guide biological construction.
-
Cancer and Aging
Mechanical forces also play roles in diseases such as cancer. Tumors grow rapidly and often disrupt the normal mechanical balance of tissues. Studying how healthy tissues use mechanical signals to maintain organization may help scientists understand how this order collapses during cancer progression.
Aging tissues also change mechanically, often becoming stiffer and more brittle. These alterations may affect how tissues respond to stress and could influence the development of age-related diseases.
The Bigger Picture
There is something deeply elegant about this idea. Nature does not avoid destruction; it integrates it into the process of creation. The same physical principles that cause cracks in dried mud or fractures in glass can, at microscopic scales, help shape living tissues.
In this sense, biology is not only chemistry but also architecture — governed by forces, stresses, and structural responses. The body develops not in spite of the laws of physics, but because of them.
The next time you see a crack spreading across dry earth or watch fabric tear along a seam, consider that similar principles may have guided the formation of the organs inside your body.
Conclusion
The study of fracture in living systems is reshaping our understanding of how bodies are built. It shows that mechanical forces in organ development play a crucial role in shaping tissues and guiding cells into the complex architectures that make life possible. What once appeared to be damage may actually be a powerful organizing principle in biology — reminding us that even breakage can be part of creation.


